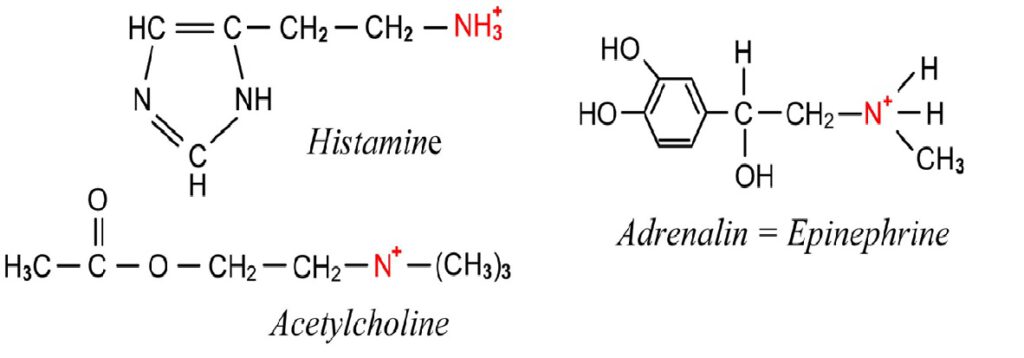
Private service
Making quantum embryos
- Our mission
- Scientific background before us
- articles
- Movies
- Conceptual illustrations
- Our scientific articles and books
- Licenses, patents and letters of recommendation
- World awards and medals
- Our centers and services
- Offices, branches and laboratories
- Land of cosmic health
- Treatment and sale of therapeutic products
- Buy a pyramid unit
- Metaverse